Article by Vivek Radhakrishnan
Article of Discussion: “A nonsurgical brain implant enabled through a cell electronic hybrid for focal neuromodulation“, Sarkar Lab, MIT
💡 Why Do Brain Implants Need a Makeover?
Electronic devices have long been implanted in the body to diagnose disease, deliver therapy, and unlock the secrets of the nervous system. Deep brain stimulators already help millions of patients with Parkinson’s disease and epilepsy. Yet every single one of these devices shares a fundamental problem: they require surgery.
Opening the skull to insert an electrode destroys tissue along the insertion track, triggers immune responses, and carries real risks of infection and hemorrhage. The implant sits rigidly in soft, dynamic brain tissue — and the brain pushes back, encapsulating the device in a glial scar that blunts its signal over time. There is a fundamental mismatch between the hard, bulky nature of conventional electronics and the soft, living world of neural tissue.
What if, instead of forcing an implant into the brain with a scalpel, we could let the body deliver it?
🧬 Can we engineer subcellular electronic devices that hitch a ride on immune cells, travel through the bloodstream, and self-implant in precise brain regions — all without a single incision?
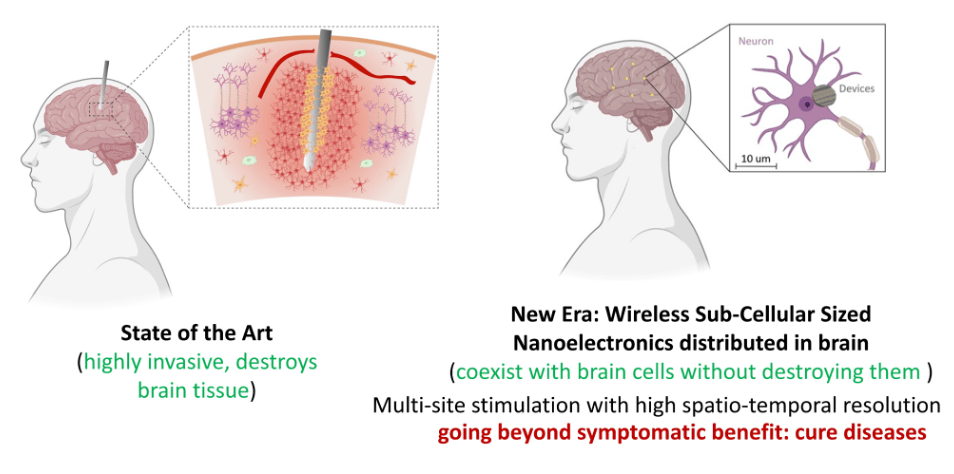
Figure 1: Conventional brain implants (left) require invasive surgery and destroy tissue along the insertion track. Subcellular wireless electronic devices (SWEDs, right) are designed to coexist with brain cells at the 10 μm scale, enabling multi-site stimulation with high spatiotemporal resolution.

🔬 Building the Device: Subcellular Wireless Electronic Devices (SWEDs)
The core innovation from the Sarkar Lab at MIT is a class of devices they call Subcellular Wireless Electronic Devices, or SWEDs. These are organic photovoltaic chips — essentially miniature solar cells — that convert near-infrared (NIR) light into electrical current. The key engineering feat is their size: just 200 nm thick and as small as 10 μm in diameter, making them genuinely smaller than many cell types in the body.
Each SWED is a three-layer sandwich:
- Anode: PEDOT:PSS, a conducting polymer that interfaces with biology
- Active layer: A binary blend of semiconducting organic polymers (donor: PCPDTBT or P3HT; acceptor: PCBM) — this is where light energy is captured and converted to electrical charge
- Cathode: Titanium, providing a stable electrical output interface
Fabrication involves conventional semiconductor techniques: spin-coating organic layers onto silicon wafers, photolithography to pattern the devices, and a clever lift-off process using a sacrificial aluminum layer dissolved by TMAH solution. A single 4-inch wafer yields approximately 33.6 million individual SWEDs. Characterization showed that even at diameters below 10 μm, these devices generate nanowatts of power — enough to stimulate neurons. Crucially, ex vivo testing through intact mouse brain tissue confirmed they can generate 0.54 nW of power even through skull and whole brain combined.
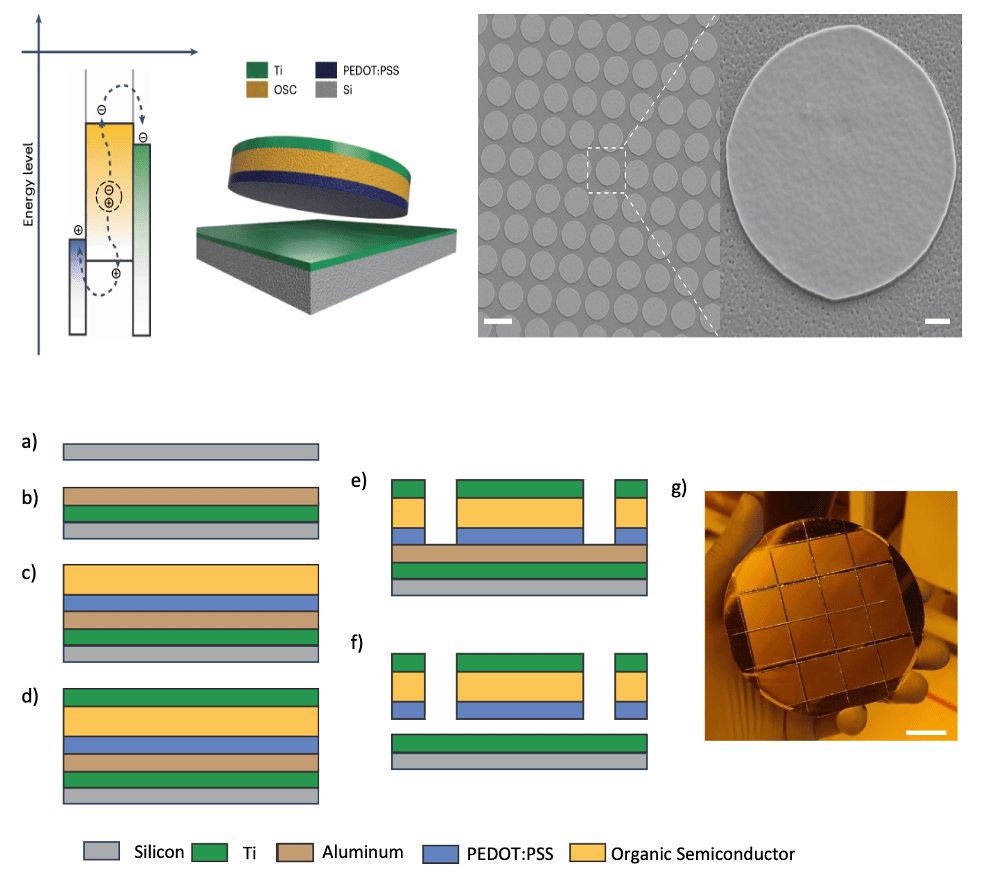
Figure 2: Schematic cross-section of a SWED showing the three-layer architecture: PEDOT:PSS anode, organic semiconductor active layer (donor: acceptor blend), and titanium cathode. (Right) Scanning electron microscopy image of fabricated SWEDs at 10 μm diameter on a silicon substrate.
🤝 The Hitchhiker Strategy: Cell-Electronics Hybrids
A SWED by itself, injected into the bloodstream, would be rapidly cleared by the immune system or become lodged in the wrong tissue. The researchers’ elegant solution was to co-opt the immune system itself: attach SWEDs to monocytes, the circulating immune cells that naturally traffic to sites of inflammation in the brain.
The attachment chemistry is elegant: the PEDOT:PSS surface of each SWED is functionalized with dibenzocyclooctyne (DBCO) groups, while the monocyte surface is modified with azide groups. These two reactive groups undergo a spontaneous, highly selective reaction called strain-promoted azide-alkyne cycloaddition — a form of bioorthogonal click chemistry — creating stable covalent bonds that lock the SWED to the cell surface without disrupting cell function.
Transmigration assays using Boyden chamber systems confirmed that 86.9% ± 0.9% of these cell-electronics hybrids remained intact and stable as the monocytes squeezed through narrow pores mimicking vessel walls. The hybrids transmigrated at a rate of 5.24 ± 0.98 mm⁻² h⁻¹ — demonstrating that attaching a device to a monocyte does not meaningfully impair the cell’s remarkable ability to navigate tight spaces.

Figure 3: Transmission electron microscopy image of a cell-electronics hybrid showing a SWED (dark disc) attached to the surface of a monocyte via bioorthogonal click chemistry, forming the biological vehicle for targeted brain delivery.

📊 Key Findings: Self-Implantation and Neuromodulation
🎯 Brain Region-Specific Delivery
The team tested autonomous delivery in mice by first inducing focal brain inflammation in the ventrolateral thalamic nucleus via stereotactic injection of lipopolysaccharide (LPS). Cell-electronics hybrids were then delivered intravenously — no surgery, just a tail vein injection. Over the next 72 hours, the monocytes followed their natural inflammatory homing signals into the brain, carrying their SWED passengers with them.
Quantification by titanium content analysis revealed approximately 14,029 ± 4,154 SWEDs successfully installed at the target brain region in the LPS + cell-electronics hybrid group — significantly more than in controls receiving SWEDs without monocyte carriers or hybrids without an inflammation target. The monocyte ‘Trojan horse’ strategy worked.
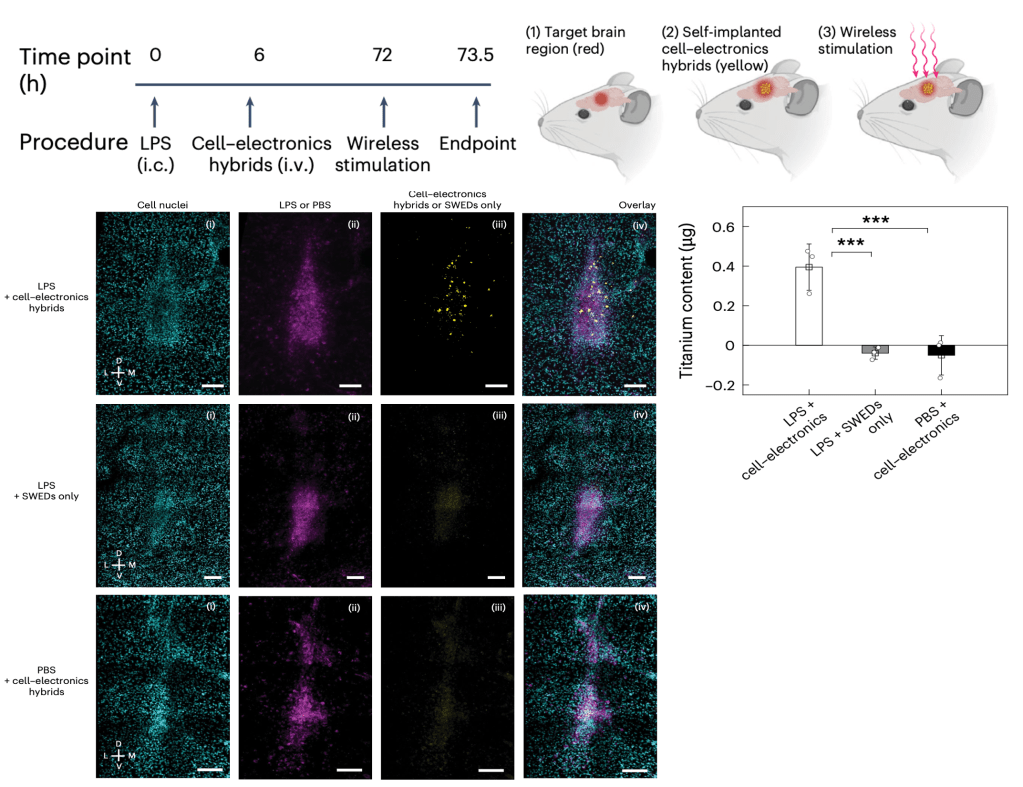
Figure 4: Autonomous self-implantation of cell-electronics hybrids in vivo. Fluorescence microscopy of coronal brain sections showing cell nuclei (cyan), LPS-induced inflammation (magenta), and SWED localization (yellow). Top row: LPS + cell-electronics hybrids show co-localization of SWEDs at the inflamed target region. Middle and bottom rows: controls (SWEDs only or hybrids without inflammation) show absent or diffuse device distribution. Bar graph shows significantly higher titanium content in the LPS + hybrids group (***p < 0.001).
⚡ Wireless Focal Neuromodulation
Once implanted, SWEDs were activated wirelessly by shining near-infrared light (792 nm) through the intact skull. In vitro patch-clamp experiments showed that a ten-pulse optical train reliably drove neurons to fire action potentials — each pulse triggering multiple spikes with precise temporal correlation to the light offset. No SWEDs, no firing.
In vivo validation used c-Fos immunohistochemistry (IHC), a standard marker for neuronal activation. Mice receiving hybrids plus wireless NIR stimulation showed 317.8 ± 80.96 c-Fos positive cells/mm² in the target region — roughly three times higher than controls with hybrids but no stimulation (107.9 ± 40.57 cells/mm²), and over four times higher than monocytes alone with stimulation (76.2 ± 64.34 cells/mm²). Spatial analysis confirmed the activation was tightly clustered around the implantation site, with c-Fos density falling off with distance from the target — a hallmark of focal stimulation.
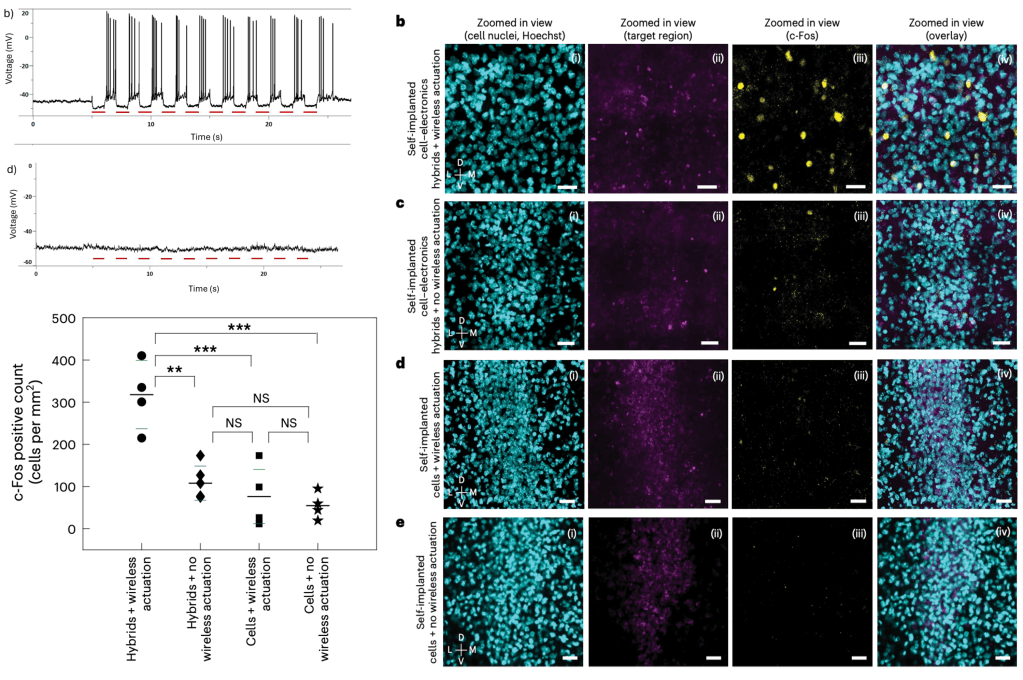
Figure 5: In vivo wireless neuromodulation by self-implanted cell-electronics hybrids.(Left, top) Patch-clamp recordings showing robust action potential firing time-locked to NIR optical stimulation pulses (red bars, 30 mW/mm²) in neurons drop-cast with SWEDs (b), but no response in SWED-free controls (d). (Left, bottom) c-Fos quantification across four groups showing significantly elevated neuronal activation only in the hybrids + wireless actuation condition (317.8 ± 80.96 cells/mm²) versus all controls (**p < 0.01, ***p < 0.001; NS = not significant). (Right, panels b–e) Fluorescence microscopy of the target brain region (ventrolateral thalamus) showing cell nuclei (cyan), target region marker (magenta), c-Fos (yellow), and overlay. Dense c-Fos signal is present only when both self-implanted hybrids and wireless actuation are combined, confirming that device delivery and optical stimulation are each necessary for focal neural activation. Scale bars: 50 µm.
🛡️ Safety and Biocompatibility
A major concern with any injectable device is safety. The team performed extensive biocompatibility assessments: complete blood counts, comprehensive serum chemistry panels, behavioral testing (open field test and novel object recognition), and histology of major organs. None revealed adverse effects from the cell-electronics hybrids.
Perhaps most reassuringly, fluorescence imaging tracking showed the hybrids were cleared from the body within 10 days post-injection. The devices do not accumulate indefinitely — a critical safety requirement. Organ distribution at day 12 showed minimal residual signal across brain, heart, kidney, liver, lungs, and spleen compared to no-hybrid controls.
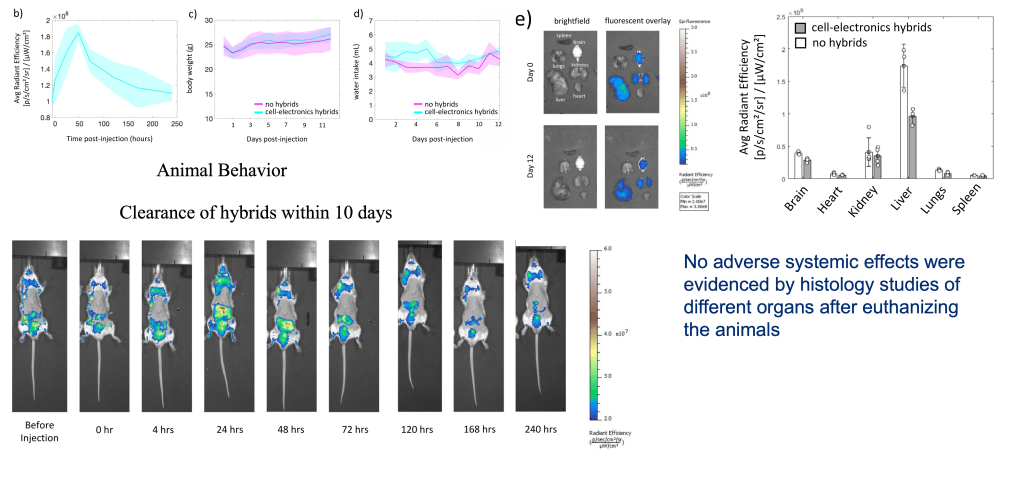
Figure 6: In vivo biocompatibility and clearance of cell-electronics hybrids. (Top) Whole-body fluorescence imaging at serial time points (0–240 hours post-injection) demonstrating progressive clearance of hybrids from the body within 10 days. (Bottom right) Organ-specific radiant efficiency at day 12 shows no significant accumulation in brain, heart, kidney, liver, lungs, or spleen compared to no-hybrid controls, confirming systemic biocompatibility.

⚠️ Limitations and Open Questions: The Road Ahead
This is a landmark proof-of-concept, but significant engineering and biology challenges remain before clinical translation is realistic:
- Power delivery depth: NIR light penetration is currently limited to approximately 2 mm of brain tissue, restricting the approach to relatively superficial targets in larger brains.
- Device lifetime and byproducts: Clearance within 10 days is shown, but the degradation byproducts of the organic semiconductors and titanium are not yet characterized.
- No recording capability: Current SWEDs are stimulation-only. A closed-loop system — one that can both sense and stimulate — would be far more therapeutically powerful.
- Device migration: Post-implantation movement from the target site remains a potential concern, particularly in dynamic tissue.
- Dependence on inflammation: The current targeting strategy requires pre-existing or artificially induced neuroinflammation — elegant for certain diseases, but limiting for others.
- Individual variability: Patient-to-patient differences in immune cell behavior and neuroinflammatory responses will require careful optimization.
Open questions worth pondering:
- Can surface engineering of monocytes or engineering of other cell types (e.g., T cells, natural killer cells) be used to target non-inflammatory brain regions?
- Could sensing capability be added — for instance, by incorporating electrochemical or optical reporters into the SWED structure?
- Is the ‘circulatronics’ platform adaptable beyond the brain — to cardiac tissue, tumors, or peripheral nerves ?

🚀 Future Vision: Circulatronics as a New Paradigm
The vision the Sarkar Lab articulates — which they call ‘Circulatronics’ — is a future where injectable, wireless, biodegradable electronics circulate through the bloodstream, hitch rides on specific cell types, self-implant at disease sites, perform their therapeutic function, and then clear away. No surgery. No permanent hardware. No scarring.
The implications span neurological diseases where existing treatments are fundamentally limited by access. Neurodegeneration, stroke, neuropathic pain, nerve injury, epilepsy — all involve spatially discrete brain regions where focal, minimally invasive modulation could be transformative. A platform that combines the targeting precision of immunology with the functional power of electronics represents something genuinely new in the history of neuromodulation.
Near-term roadmap items include integrating sensing and closed-loop control using CMOS-compatible nanoelectronics, expanding modalities beyond optical harvesting (e.g., ultrasound or radio frequency), and engineering degradable or cleavable linkers to precisely tune device lifetime for chronic versus transient applications.
It is worth noting the elegant convergence this work represents: it merges semiconductor fabrication, organic chemistry, immunology, and neuroscience into a single therapeutic platform. Each of those fields has been maturing independently for decades. The insight here is that their intersection — a cell-electronic hybrid — unlocks capabilities none could achieve alone.
If this article excited you, consider liking, sharing, and following our journey at Grow Your Tissues.
References:
Article of discussion:
- Yadav, S., Lee, R.X., Kajale, S.N. et al. A nonsurgical brain implant enabled through a cell–electronics hybrid for focal neuromodulation. Nat Biotechnol (2025). https://doi.org/10.1038/s41587-025-02809-3

Leave a comment