Article by Vivek Radhakrishnan
Article of Discussion: “Gastruloids Enable Modeling of the Earliest Stages of Human Cardiac and Hepatic Vascularization”, Wu Lab, Stanford University
💡 Why do we need vascularized organoids?
Human pluripotent stem cells (hPSCs) can become anything — neurons, cardiomyocytes, hepatocytes — the ultimate biological blank slate. In recent years, these cells have been coaxed into forming organoids — miniature, 3D structures that mimic early organs. Yet, they all share one fatal flaw: the lack of blood vessels.
Without a vascular network to deliver oxygen and nutrients, organoids grow sluggishly, their cores die from hypoxia, and their ability to mimic real human organs remains limited. This “vascularization bottleneck” has kept organoid models immature and nonfunctional, especially for organs like the heart and liver, where blood flow is everything.
But studying the earliest stages of human vascular development in embryos is nearly impossible due to ethical and technical constraints. So here’s the million-dollar question:
🧬 Can we recreate the first steps of blood vessel formation in the lab, using gastruloids derived from hPSCs? And utilize this knowledge to develop de novo vasculature?
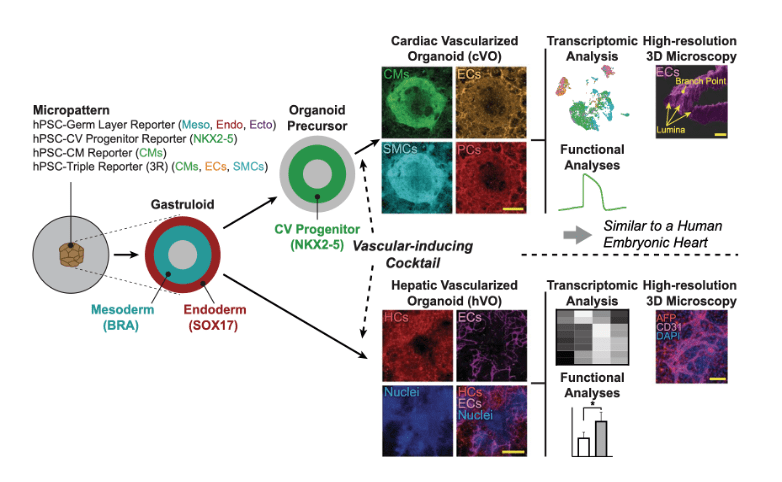

🧫 Building the Model: Gastruloids as a Window Into Early Life
At the heart of this study are human pluripotent stem cells (hPSCs)—the blank canvas cells capable of becoming any cell type in the human body.
The team used micropatterning techniques – creating precise, micrometer-scale patterns on cell culture surfaces to control cell adhesion, organization, and behavior – to grow these hPSCs into organized 2D colonies that self-organized into gastruloids. They are structures that mimic the earliest embryonic stages (Carnegie stages 9–10). These gastruloids contained the three germ layers—ectoderm, mesoderm, and endoderm—and provided a scaffold for de novo vascularization.
To guide their fate, the researchers created an intricate temporal “growth factor cocktail”, mimicking the natural developmental timeline:
- Days 0–3: Mesoderm induction using CHIR99021 (Wnt activator), FGF2, and IWR-1 (Wnt inhibitor).
- Days 5–16: Vasculogenesis and angiogenesis using VEGF, FGF2, SB431542 (TGF-β inhibitor), and angiopoietins.
- Days 7–16: Smooth muscle recruitment and vessel maturation via PDGF-BB and TGF-β1
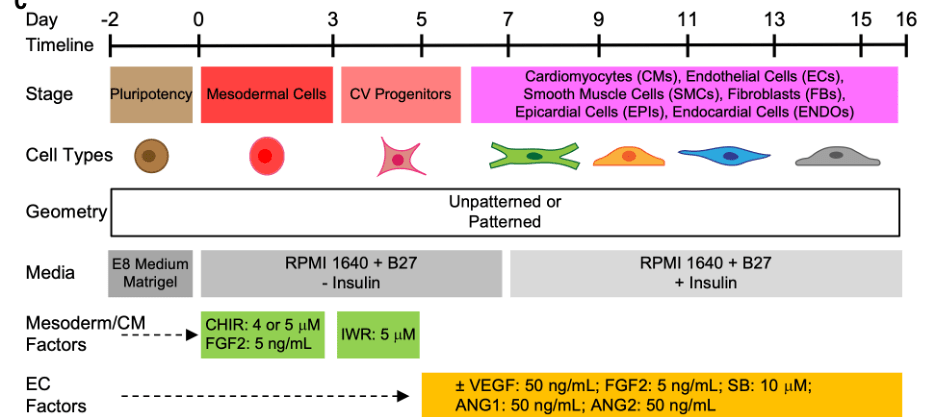
Figure 1: Differentiation protocol
Among the 34 conditions tested, Condition 32 emerged as the winning recipe—yielding a tri-lineage mix of cardiomyocytes (CMs), endothelial cells (ECs), and smooth muscle cells (SMCs) in a single, structured organoid.
The result? The first-ever cardiac vascularized organoids (cVOs) formed entirely in vitro.

📊 Key Findings: Progress in Vascularized Gastruloids
The study yielded significant insights and outcomes in structural mimicry, biological modeling, and functional vascular formation. Below are the notable observations:
🏗️ Structural Mimicry: Embryonic Integrity in a Controlled Model
- Dynamic Self-Organization : Gastruloids generated by micropatterning techniques successfully replicated human embryonic stages (Carnegie stages 9–10), a crucial early time point in development where foundational structures emerge.
- Tri-lineage Differentiation : By applying the Condition 32 growth-factor cocktail, researchers achieved co-differentiation into three critical cell types—cardiomyocytes, endothelial cells, and smooth muscle cells—all within a single structured organoid.
- Scaffold Formation : Cellular alignment and architecture were preserved, demonstrating that gastruloids could mimic embryonic tissue organization accurately.
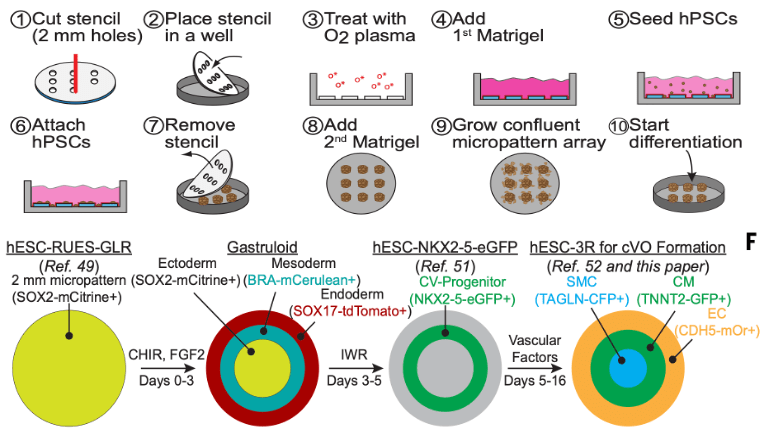
Figure 2: Image depicting the micro patterning techniques used for gastruloid formation. A visual representation of how hPSCs are structured into gastruloids, highlighting tri-lineage distribution in the gastruloid (cardiomyocytes, endothelial cells, and smooth muscle cells)
🧬 Biological Mimicry: Multi-lineage Success
- Vascular Emergence : Endothelial cells successfully clustered and organized into primitive vascular structures, illustrating the gastruloids’ potential to model vasculogenesis.
- Cellular Crosstalk : Smooth muscle cells were recruited effectively into vascular networks, emulating natural processes of vessel maturation observed during embryonic development.
- Mimicked Cardiac Microenvironments : Integration of cardiomyocytes into vascularized gastruloids allowed researchers to establish heart-like cellular patterns and dynamics in vitro for the first time
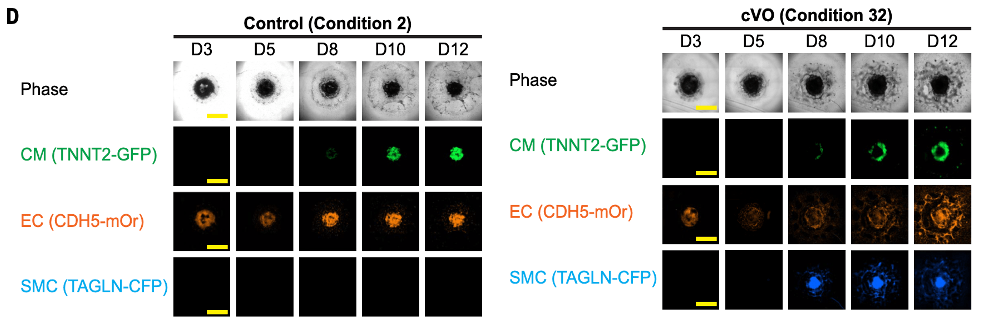
Figure 3: Visual comparison of native cardiac cellular structures versus gastruloid-derived structures upon subjection to the differentiation cocktail being screened. Note the differences in the simultaneous yield of CMs, ECs, and SMCs, forming cardiac vascularized organoids (cVOs).
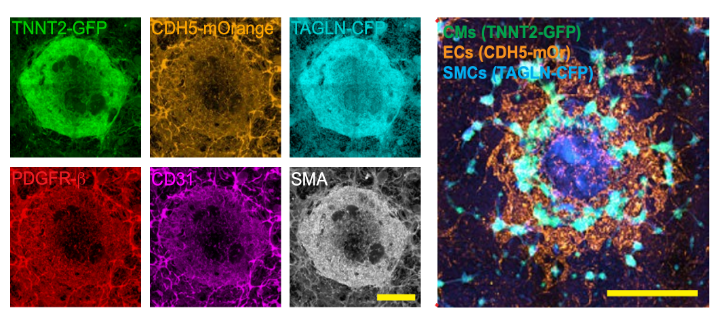
Figure 4: Fluorescent microscopy imaging showing vascular structures (EC recruitment, Smooth muscle integration and tissue layering)
⚙️ Functional Capability: First Generation Cardiovascularized Organoids
- Cardiac Vascularized Organoids (cVOs) : The study reported the creation of structured, multi-cellular organoids capable of supporting primary cardiac vascular connection pathways. Condition 32 reliably facilitated vascular formation processes, resulting in unprecedented cellular interactions within controlled environments.
- Enabled Smooth Muscle Integration : The organoids achieved vessel maturation, as smooth muscle cells were effectively recruited to form stable structures—a key requirement for mimicking vascular systems.
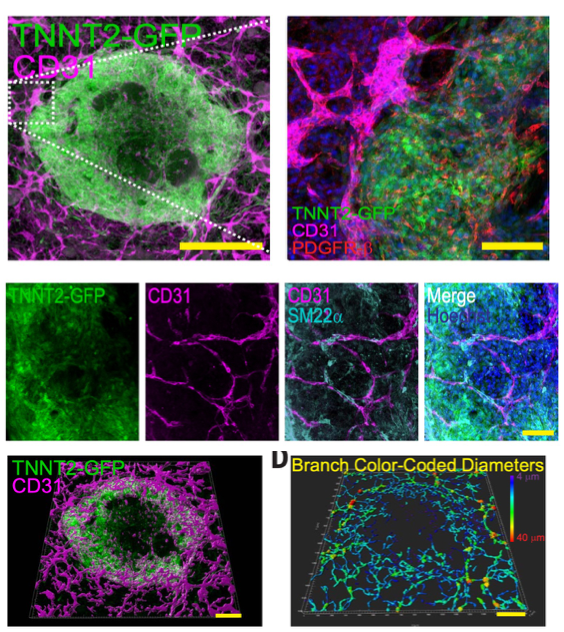
Figure 2: Representative fluorescence microscopy images showing gastruloid-derived vascularized cardiac organoids (cVOs).(A) TNNT2-GFP highlights cardiomyocytes, while CD31 marks endothelial cells involved in forming organized vascular networks.(B) Merged images demonstrate the integration of PDGFRβ and SM22α positive smooth muscle cells into vascular structures.(C) Multi-channel views illustrate co-localization of cardiomyocytes (TNNT2-GFP), endothelial cells (CD31), and smooth muscle markers, indicating tri-lineage differentiation and vascular formation. (D) 3D reconstruction of vascular networks with branch diameter mapping highlights geometric complexity and structural organization.
🔄 Extension of Concept
Additionally, the study extended its approach to model hepatic vascularization, demonstrating gastruloids’ universal potential in differentiation. This proof-of-concept marks the versatility of gastruloids as a system for modeling vascular development across multiple organ systems, including the heart and liver.
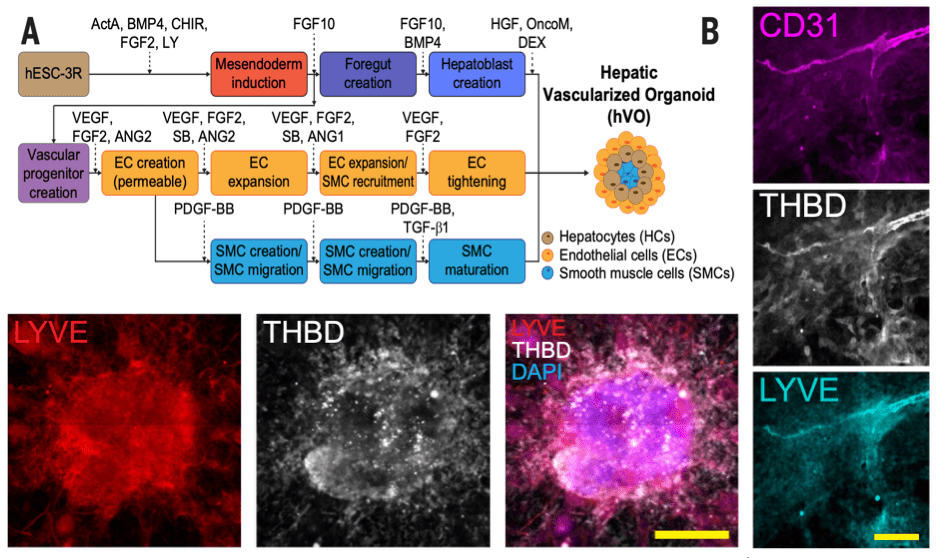
Figure 3: Extension to create Hepatic Vascularized Organoids (A) Schematic representation of growth-factor-guided differentiation showing the stepwise process for creating hepatic vascularized organoids (hVOs). Human embryonic stem cells (hESCs) were directed through mesoderm induction, foregut formation, and hepatoblast creation, while endothelial cells (ECs), smooth muscle cells (SMCs), and hepatocytes (HCs) were simultaneously recruited and matured to mimic native hepatic tissue. (B) Fluorescence images demonstrating vascularization within hVOs. CD31 marks endothelial cells, THBD demonstrates thrombomodulin expression, and LYVE indicates lymphatic vessel endothelial marker integration. Merged images (LYVE, THBD, DAPI) highlight the spatial organization and cellular interactions across vascular networks within the organoid.
⚠️ Limitations and Next Steps: Bridging Remaining Gaps
📈 Functional Testing and Observations:
While the organoids offer groundbreaking promise, certain gaps in validation emerged during testing:
- Vascular systems appeared stable in structure but lacked long-term assessments under dynamic conditions (increased flow, pressure, or shear stress).
- Functional assays for nutrient exchange, epithelial barrier formation, and cardiac rhythm synchronization were not conducted in this phase, leaving more physiological validations pending for future iterations.
- Limited testing environments, such as cultured systems and isolated static setups, left questions about performance under active cardiac pulsations unresolved.
- Limited Immune Component: A key missing element is hematopoietic/immune cells (macrophages, etc.), which play active roles in development and disease.
Open Questions:
- Can we use this platform to model vascular-centric diseases like Hypoplastic Left Heart Syndrome?
- What factors are required to push these organoids to a more mature state?
- As shown that vascularization was organ specific, could this when applied to brain organoids, be able to develop de novo blood brain barrier ?
This results section showcases gastruloids as an exciting new tool for developmental modeling, offering a scaffold for exploring early human cardiac and vascular interactions under regulated experimental conditions. Further refinement may help address current gaps and uncover the full regenerative and functional potential of Model 32 gastruloids.
🚀 Future Vision: A Window Into Human Heart Development
As gastruloids give researchers a closer glimpse at early life development in vitro, they illuminate pathways toward functional organoid engineering. These efforts push the boundaries of regenerative medicine and propose visions where miniature lab-created cardiac networks become stepping stones for real-world applications—treatments for cardiovascular failure, mechanisms of heart repair, and even bypass surgical grafts.
Through continued collaboration across developmental biology, stem cell technology, and computational modeling, gastruloids bring us closer to understanding the cradle of human life and translate stem cell potential into lifesaving innovations.
If this article excited you, consider liking, sharing, and following our journey at Grow Your Tissues.
References:
References:
Article of discussion:
- Oscar J. Abilez et al.,Gastruloids enable modeling of the earliest stages of human cardiac and hepatic vascularization. Science 388, eadu9375(2025). DOI:10.1126/science.adu9375

Leave a comment